Laboratory for Food Safety (Maisons-Alfort and Boulogne-sur-Mer)
Director: Laurent Laloux
Deputy Scientific Director: Jacques-Antoine Hennekinne
Deputy Director for General Affairs: Catherine Lambert
Addresses:
Maisons-Alfort: 14 rue Pierre & Marie Curie 94701 Maisons-Alfort Cedex - Tel.: +33 (0)1 49 77 13 50
Boulogne-sur-Mer: 6 boulevard Bassin Napoléon 62200 Boulogne-sur-Mer - Tel.: +33 (0)3 21 99 25 00
Email: contact.LSAL@anses.fr
Employees: around 140 spread out across 10 units and one technology platform
The Laboratory for Food Safety focuses on biological and chemical hazards that can affect food safety and quality. It has two sites, in Maisons-Alfort and Boulogne-sur-Mer.
“Food safety” refers to efforts to guarantee the safety of food products, i.e. ensure that their consumption will not have negative health consequences. The laboratory’s scientific activities aim to enrich knowledge on the types of contaminants or “hazards” found in food, understand their mechanisms of action and effects on health, and help improve policies for protecting consumer health.
The Maisons-Alfort site has 120 employees, specialising in food safety, while the Boulogne-sur-Mer site has over 20 staff members, dedicated exclusively to fishery and aquaculture products.
The laboratory's missions
The Laboratory for Food Safety provides public decision-makers with general scientific expertise on the quality and safety of foodstuffs, as well as more specific expertise relating to certain processing sectors.
The laboratory's work focuses on biological (bacteria, viruses, parasites) and chemical (toxins, metal trace elements, pesticides, organic pollutants, histamine) hazards that are found or likely to be found in foods, especially during their distribution and consumption:
- it produces knowledge on the identification of food hazards;
- it develops methods for detecting, characterising and quantifying them;
- it analyses their causes and development factors;
- it helps monitor them, particularly to report the emergence or re-emergence of certain organisms and compounds;
- it helps standardise and draft regulations for specific methods and criteria governing these hazards;
- it helps assess the related risks by contributing to quantitative risk analyses, and by providing data on the prevalence of a biological hazard or the occurrence of a chemical hazard;
- it examines how practices and processes influence the expression of food hazards and specifically focuses on the effectiveness of control measures undertaken throughout a food's lifetime (from farm to fork) and on quality indicators related to hygiene.
The laboratory also performs analyses for health authorities and professionals in relation to fishery products (fish, shellfish, crustaceans) in Boulogne-sur-Mer and in relation to all types of foodstuffs in Maisons-Alfort, in the framework of the Central Laboratory for Veterinary Services, which carries out official analyses for the Departmental Directorates for the Protection of Populations (DDPPs) in the Paris region.
The laboratory hosts the national “IdentyPath” technology platform for the identification and typing of pathogens using high- and medium-throughput PCR.
Main biological contaminants studied:
- Salmonella;
- Pathogenic E. coli;
- Coagulase-positive staphylococci;
- Listeria monocytogenes;
- Bacillus cereus;
- Vibrio;
- Clostridium;
- Enteric viruses (HAV, HEV, noroviruses, enteroviruses);
- Fish parasites, in particular Anisakidae.
Main chemical contaminants studied:
- pesticides (organochlorines, organophosphates, pyrethroids, carbamates, etc.)
- metal (lead, mercury, cadmium, copper, etc.) and non-metal (arsenic, selenium, etc.) trace elements, nanomaterials, and minerals (sodium, potassium, calcium, magnesium, etc.)
- bacterial toxins (Staphylococcus aureus and Bacillus cereus toxins)
- marine biotoxins (lipophilic toxins, domoic acid, saxitoxins, pinnatoxins, ciguatoxins, etc.)
- cyanotoxins (microcystins, etc.)
- biogenic amines (histamine, cadaverine, putrescine, etc.)
- nano- and microplastics.
To know more about the Laboratory
>> See the leaflet (PDF)
Latest laboratory news
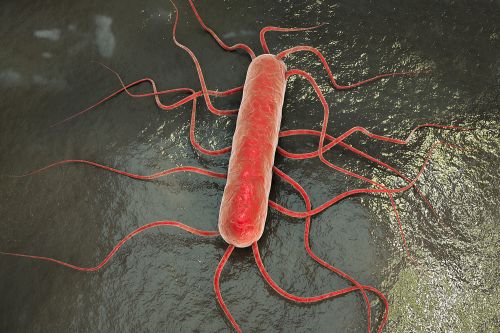
Listeria monocytogenes: a surprisingly adaptive bacterium
Listeria monocytogenes is a ubiquitous bacterium found in food, livestock and wild animals, soil, water and vegetation. The European ListAdapt project focused on the ability of strains of this bacterium to adapt to these different environments. It found that this ability is independent of the strains' environments of origin or their belonging to a given sub-group.
The laboratory's units
The laboratory has 10 units, one technological platform, three scientific departments and six support services, spread out across two sites: Maisons-Alfort and Boulogne-sur-Mer.
- Salmonella and Listeria (SEL) unit;
- Staphylococcus, Bacillus, Clostridium (SBCL) unit;
- Pathogenic E. coli unit (Colipath);
- Central Laboratory for Veterinary Services (LCSV) unit;
- Enteric Viruses (VE) unit;
- Bacteriology and Parasitology of Fishery and Aquaculture Products (B3PA) unit;
- Pesticides and Marine Biotoxins (PBM) unit;
- Metal Trace Elements and Minerals (ET2M) unit;
- Physico-Chemistry of Fishery and Aquaculture Products (PCPPA) unit;
- Biochemistry of Aquatic Products (BPA) unit, in partnership with the University of the Littoral-Côte d’Opale;
- National Platform for the Identification and Typing of Pathogens (IdentyPath).
Partnerships
Scientific partnerships are a high priority for the laboratory, with the main objective of developing complementary skills and analytical approaches with external partners.
In this context, long-term regional partnerships have been created, in particular with universities such as the University of the Littoral-Côte d’Opale (ULCA) in the Hauts-de-France region and University of Paris-Est-Créteil (UPEC) in the Île-de-France region. Arrangements in the form of federative research structures (SFRs) or university schools of research (EURs) are being considered with these partners.
Internationally, the laboratory is involved in strategic partnerships between ANSES or the French State and various European agencies, including its counterparts from the German Federal Institute for Risk Assessment, the Technical University of Denmark, Sciensano in Belgium, and the Italian National Institute of Health (ISS). Outside of Europe, emphasis is placed on partnerships with large North American organisations (United States, Canada); partnerships with India, China and South-east Asia are also being studied.
National partners
- Network for food technology institutes (ACTIA), Network for agricultural technology institutes (ACTA)
- University hospitals
- Food technology institutes
- French National Centre for Scientific Research (CNRS)
- French Alternative Energies and Atomic Energy Commission (CEA)
- Hauts-de-France Regional Council
- Île-de-France Regional Council
- Departmental Directorates for the Protection of Populations
- Agricultural schools (AgroParisTech)
- Veterinary schools
- Trade federations
- French Research Institute for Exploitation of the Sea (Ifremer)
- French National Research Institute for Agriculture and the Environment (INRAE)
- Institut Pasteur (Paris and Lille)
- National Metrology and Testing Laboratory (LNE)
- Ministry of Agriculture and Food (Directorate General for Food)
- Ministry of Solidarity and Health (Directorate General for Health)
- French Natural History Museum
- Aquimer competitiveness cluster
- Santé publique France (SPF)
- Joint Laboratory Service (SCL)
- Universities (Paris-Est Sup, Paris Saclay, Lille Nord de France, Littoral-Côte d’Opale, etc.)
International partnerships
- European Food Safety Authority (EFSA)
- Centres for Disease Control and Prevention (CDC, ECDC)
- Canadian Food Inspection Agency (CFIA)
- Italian National Institute of Health (ISS)
- European Committee for Standardization (CEN)
- European Commission (DG Santé)
- German Federal Institute for Risk Assessment (BfR)
- Finnish Food Safety Authority (EVIRA)
- Italian health authority and research organization for animal health and food safety (Italy)
- Joint Research Centre (Geel, Belgium and Ispra, Italy)
- National Institute for Public Health and the Environment (RIVM, Netherlands)
- National Institute of Standards and Technology (NIST, USA)
- Swedish National Veterinary Institute (SVA)
- International Organization for Standardization (ISO, Switzerland)
- Public Health Agency of Canada (PHAC)
- Public Health England (PHE)
- Networks of National Reference Laboratories of European countries
- Technical University of Denmark (DTU)
- University of Oviedo (Spain)
- Animal & Plant Health Agency (APHA, England)
The laboratory's services
Analytical services
Addresses and opening hours for sample deliveries:
Boulogne-sur-Mer site
Opening hours: 9:00 am - 12:30 pm / 2:00 - 5:00 pm
6 Boulevard Bassin Napoléon, 62200 Boulogne-sur-Mer
Maisons-Alfort site
Opening hours: 9:00 am - 4:30 pm
14 rue Pierre et Marie Curie, 94701 Maisons-Alfort Cedex
The analytical services offered by the Laboratory for Food Safety and their implementation conditions are listed in the Service catalogue. You can also view the rates and the general terms and conditions of sale.
The units of the Laboratory for Food Safety (LCSV, PBM pesticides team, VE) are equipped with analysis management software and can send you analysis reports electronically, instead of by post. This shortens delivery lead times and facilitates transmission. To benefit from this service, please send us a signed copy of the agreement on proof.
The laboratory has been accredited by COFRAC according to the NF EN ISO/CEI 17025 standard under the numbers 1-2246 (Maisons-Alfort) and 1-5959 (Boulogne-sur-Mer) for the tests defined in the accreditation scope (available on the COFRAC website). The use of the laboratory's accreditation label by clients and stakeholders is not permitted outside of documents issued by the laboratory in their full form (e.g. test reports).
ILPT organisation
The inter-laboratory proficiency tests organised by the Laboratory for Food Safety and the registration conditions are available on the LEILA platform.
The laboratory (Maisons-Alfort site) has been accredited by COFRAC according to the NF EN ISO/CEI 17043 standard under the number 1-6993 for the organisation of inter-laboratory proficiency testing as defined in the accreditation scope (available on the COFRAC website) and in the detailed accreditation scope (PDF in French). The use of the laboratory's accreditation label by clients and stakeholders is not allowed outside the documents issued by the laboratory in their full form (e.g. Inter-laboratory proficiency testing reports).